| Home | Sources Directory | News Releases | Calendar | Articles | | Contact | |
Lipid

Lipids are a broad group of naturally occurring molecules which includes fats, waxes, sterols, fat-soluble vitamins (such as vitamins A, D, E and K), monoglycerides, diglycerides, phospholipids, and others. The main biological functions of lipids include energy storage, as structural components of cell membranes, and as important signaling molecules.
Lipids may be broadly defined as hydrophobic or amphiphilic small molecules; the amphiphilic nature of some lipids allows them to form structures such as vesicles, liposomes, or membranes in an aqueous environment. Biological lipids originate entirely or in part from two distinct types of biochemical subunits or "building blocks": ketoacyl and isoprene groups.[4] Using this approach, lipids may be divided into eight categories: fatty acyls, glycerolipids, glycerophospholipids, sphingolipids, saccharolipids and polyketides (derived from condensation of ketoacyl subunits); and sterol lipids and prenol lipids (derived from condensation of isoprene subunits).
Although the term lipid is sometimes used as a synonym for fats, fats are a subgroup of lipids called triglycerides. Lipids also encompass molecules such as fatty acids and their derivatives (including tri-, di-, and monoglycerides and phospholipids), as well as other sterol-containing metabolites such as cholesterol.[5] Although humans and other mammals use various biosynthetic pathways to both break down and synthesize lipids, some essential lipids cannot be made this way and must be obtained from the diet.
Contents |
[edit] Categories of lipids
[edit] Fatty acyls
Fatty acyls, a generic term for describing fatty acids, their conjugates and derivatives, are a diverse group of molecules synthesized by chain-elongation of an acetyl-CoA primer with malonyl-CoA or methylmalonyl-CoA groups in a process called fatty acid synthesis.[6][7] They are made of a hydrocarbon chain that terminates with a carboxylic acid group; this arrangement confers the molecule with a polar, hydrophilic end, and a nonpolar, hydrophobic end that is insoluble in water. The fatty acid structure is one of the most fundamental categories of biological lipids, and is commonly used as a building block of more structurally complex lipids. The carbon chain, typically between four to 24 carbons long,[8] may be saturated or unsaturated, and may be attached to functional groups containing oxygen, halogens, nitrogen and sulfur. Where a double bond exists, there is the possibility of either a cis or trans geometric isomerism, which significantly affects the molecule's molecular configuration. Cis-double bonds cause the fatty acid chain to bend, an effect that is more pronounced the more double bonds there are in a chain. This in turn plays an important role in the structure and function of cell membranes.[9] Most naturally occurring fatty acids are of the cis configuration, although the trans form does exist in some natural and partially hydrogenated fats and oils.[10]
Examples of biologically important fatty acids are the eicosanoids, derived primarily from arachidonic acid and eicosapentaenoic acid, which include prostaglandins, leukotrienes, and thromboxanes. Other major lipid classes in the fatty acid category are the fatty esters and fatty amides. Fatty esters include important biochemical intermediates such as wax esters, fatty acid thioester coenzyme A derivatives, fatty acid thioester ACP derivatives and fatty acid carnitines. The fatty amides include N-acyl ethanolamines, such as the cannabinoid neurotransmitter anandamide.[11]
[edit] Glycerolipids
Glycerolipids are composed mainly of mono-, di- and tri-substituted glycerols,[12] the most well-known being the fatty acid esters of glycerol (triacylglycerols), also known as triglycerides. In these compounds, the three hydroxyl groups of glycerol are each esterified, usually by different fatty acids. Because they function as a food store, these lipids comprise the bulk of storage fat in animal tissues. The hydrolysis of the ester bonds of triacylglycerols and the release of glycerol and fatty acids from adipose tissue is called fat mobilization.[13]
Additional subclasses of glycerolipids are represented by glycosylglycerols, which are characterized by the presence of one or more sugar residues attached to glycerol via a glycosidic linkage. Examples of structures in this category are the digalactosyldiacylglycerols found in plant membranes[14] and seminolipid from mammalian sperm cells.[15]
[edit] Glycerophospholipids
Glycerophospholipids, also referred to as phospholipids, are ubiquitous in nature and are key components of the lipid bilayer of cells, as well as being involved in metabolism and cell signaling. Neural tissue (including the brain) contains relatively high amounts of glycerophospholipids, and alterations in their composition has been implicated in various neurological disorders.[16] Glycerophospholipids may be subdivided into distinct classes, based on the nature of the polar headgroup at the sn-3 position of the glycerol backbone in eukaryotes and eubacteria, or the sn-1 position in the case of archaebacteria.[17]

Examples of glycerophospholipids found in biological membranes are phosphatidylcholine (also known as PC, GPCho or lecithin), phosphatidylethanolamine (PE or GPEtn) and phosphatidylserine (PS or GPSer). In addition to serving as a primary component of cellular membranes and binding sites for intra- and intercellular proteins, some glycerophospholipids in eukaryotic cells, such as phosphatidylinositols and phosphatidic acids are either precursors of, or are themselves, membrane-derived second messengers.[18] Typically, one or both of these hydroxyl groups are acylated with long-chain fatty acids, but there are also alkyl-linked and 1Z-alkenyl-linked (plasmalogen) glycerophospholipids, as well as dialkylether variants in archaebacteria.[19]
[edit] Sphingolipids
Sphingolipids are a complex family of compounds[20] that share a common structural feature, a sphingoid base backbone that is synthesized de novo from the amino acid serine and a long-chain fatty acyl CoA, then converted into ceramides, phosphosphingolipids, glycosphingolipids and other compounds. The major sphingoid base of mammals is commonly referred to as sphingosine. Ceramides (N-acyl-sphingoid bases) are a major subclass of sphingoid base derivatives with an amide-linked fatty acid. The fatty acids are typically saturated or mono-unsaturated with chain lengths from 16 to 26 carbon atoms.[21]

The major phosphosphingolipids of mammals are sphingomyelins (ceramide phosphocholines),[22] whereas insects contain mainly ceramide phosphoethanolamines[23] and fungi have phytoceramide phosphoinositols and mannose-containing headgroups.[24] The glycosphingolipids are a diverse family of molecules composed of one or more sugar residues linked via a glycosidic bond to the sphingoid base. Examples of these are the simple and complex glycosphingolipids such as cerebrosides and gangliosides.
[edit] Sterol lipids
Sterol lipids, such as cholesterol and its derivatives, are an important component of membrane lipids,[25] along with the glycerophospholipids and sphingomyelins. The steroids, all derived from the same fused four-ring core structure, have different biological roles as hormones and signaling molecules. The eighteen-carbon (C18) steroids include the estrogen family whereas the C19 steroids comprise the androgens such as testosterone and androsterone. The C21 subclass includes the progestogens as well as the glucocorticoids and mineralocorticoids.[26] The secosteroids, comprising various forms of vitamin D, are characterized by cleavage of the B ring of the core structure.[27] Other examples of sterols are the bile acids and their conjugates,[28] which in mammals are oxidized derivatives of cholesterol and are synthesized in the liver. The plant equivalents are the phytosterols, such as î�-sitosterol, stigmasterol, and brassicasterol; the latter compound is also used as a biomarker for algal growth.[29] The predominant sterol in fungal cell membranes is ergosterol.[30]
[edit] Prenol lipids
Prenol lipids are synthesized from the 5-carbon precursors isopentenyl diphosphate and dimethylallyl diphosphate that are produced mainly via the mevalonic acid (MVA) pathway.[31] The simple isoprenoids (linear alcohols, diphosphates, etc.) are formed by the successive addition of C5 units, and are classified according to number of these terpene units. Structures containing greater than 40 carbons are known as polyterpenes. Carotenoids are important simple isoprenoids that function as antioxidants and as precursors of vitamin A.[32] Another biologically important class of molecules is exemplified by the quinones and hydroquinones, which contain an isoprenoid tail attached to a quinonoid core of non-isoprenoid origin.[33] Vitamin E and vitamin K, as well as the ubiquinones, are examples of this class. Prokaryotes synthesize polyprenols (called bactoprenols) in which the terminal isoprenoid unit attached to oxygen remains unsaturated, whereas in animal polyprenols (dolichols) the terminal isoprenoid is reduced.[34]
[edit] Saccharolipids
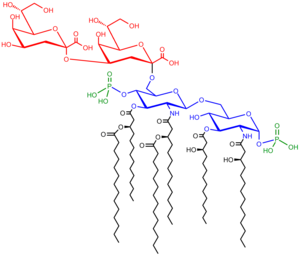
Saccharolipids describe compounds in which fatty acids are linked directly to a sugar backbone, forming structures that are compatible with membrane bilayers. In the saccharolipids, a monosaccharide substitutes for the glycerol backbone present in glycerolipids and glycerophospholipids. The most familiar saccharolipids are the acylated glucosamine precursors of the Lipid A component of the lipopolysaccharides in Gram-negative bacteria. Typical lipid A molecules are disaccharides of glucosamine, which are derivatized with as many as seven fatty-acyl chains. The minimal lipopolysaccharide required for growth in E. coli is Kdo2-Lipid A, a hexa-acylated disaccharide of glucosamine that is glycosylated with two 3-deoxy-D-manno-octulosonic acid (Kdo) residues.[35]
[edit] Polyketides
Polyketides are synthesized by polymerization of acetyl and propionyl subunits by classic enzymes as well as iterative and multimodular enzymes that share mechanistic features with the fatty acid synthases. They comprise a large number of secondary metabolites and natural products from animal, plant, bacterial, fungal and marine sources, and have great structural diversity.[36][37] Many polyketides are cyclic molecules whose backbones are often further modified by glycosylation, methylation, hydroxylation, oxidation, and/or other processes. Many commonly used anti-microbial, anti-parasitic, and anti-cancer agents are polyketides or polyketide derivatives, such as erythromycins, tetracyclines, avermectins, and antitumor epothilones.[38]
[edit] Biological functions
[edit] Membranes
Eukaryotic cells are compartmentalized into membrane-bound organelles which carry out different biological functions. The glycerophospholipids are the main structural component of biological membranes, such as the cellular plasma membrane and the intracellular membranes of organelles; in animal cells the plasma membrane physically separates the intracellular components from the extracellular environment. The glycerophospholipids are amphipathic molecules (containing both hydrophobic and hydrophilic regions) that contain a glycerol core linked to two fatty acid-derived "tails" by ester linkages and to one "head" group by a phosphate ester linkage. While glycerophospholipids are the major component of biological membranes, other non-glyceride lipid components such as sphingomyelin and sterols (mainly cholesterol in animal cell membranes) are also found in biological membranes.[39] In plants and algae, the galactosyldiacylglycerols,[40] and sulfoquinovosyldiacylglycerol,[14] which lack a phosphate group, are important components of membranes of chloroplasts and related organelles and are the most abundant lipids in photosynthetic tissues, including those of higher plants, algae and certain bacteria.
Bilayers have been found to exhibit high levels of birefringence which can be used to probe the degree of order (or disruption) within the bilayer using techniques such as dual polarisation interferometry
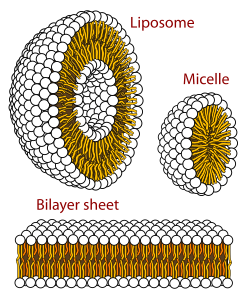
A biological membrane is a form of lipid bilayer. The formation of lipid bilayers is an energetically preferred process when the glycerophospholipids described above are in an aqueous environment.[41] In an aqueous system, the polar heads of lipids align towards the polar, aqueous environment, while the hydrophobic tails minimize their contact with water and tend to cluster together, forming a vesicle; depending on the concentration of the lipid, this biophysical interaction may result in the formation of micelles, liposomes, or lipid bilayers. Other aggregations are also observed and form part of the polymorphism of amphiphile (lipid) behavior. Phase behavior is an area of study within biophysics and is the subject of current academic research.[42][43] Micelles and bilayers form in the polar medium by a process known as the hydrophobic effect.[44] When dissolving a lipophilic or amphiphilic substance in a polar environment, the polar molecules (i.e., water in an aqueous solution) become more ordered around the dissolved lipophilic substance, since the polar molecules cannot form hydrogen bonds to the lipophilic areas of the amphiphile. So in an aqueous environment, the water molecules form an ordered "clathrate" cage around the dissolved lipophilic molecule.[45]
[edit] Energy storage
Triacylglycerols, stored in adipose tissue, are a major form of energy storage in animals. The adipocyte, or fat cell, is designed for continuous synthesis and breakdown of triacylglycerols, with breakdown controlled mainly by the activation of hormone-sensitive enzyme lipase.[46] The complete oxidation of fatty acids provides high caloric content, about 9 kcal/g, compared with 4 kcal/g for the breakdown of carbohydrates and proteins. Migratory birds that must fly long distances without eating use stored energy of triacylglycerols to fuel their flights.[47]
[edit] Signaling
In recent years, evidence has emerged showing that lipid signaling is a vital part of the cell signaling.[48] Lipid signaling may occur via activation of G protein-coupled or nuclear receptors, and members of several different lipid categories have been identified as signaling molecules and cellular messengers.[49] These include sphingosine-1-phosphate, a sphingolipid derived from ceramide that is a potent messenger molecule involved in regulating calcium mobilization,[50] cell growth, and apoptosis;[51] diacylglycerol (DAG) and the phosphatidylinositol phosphates (PIPs), involved in calcium-mediated activation of protein kinase C;[52] the prostaglandins, which are one type of fatty-acid derived eicosanoid involved in inflammation and immunity;[53] the steroid hormones such as estrogen, testosterone and cortisol, which modulate a host of functions such as reproduction, metabolism and blood pressure; and the oxysterols such as 25-hydroxy-cholesterol that are liver X receptor agonists.[54]
[edit] Other functions
The "fat-soluble" vitamins (A, D, E and K) ' which are isoprene-based lipids ' are essential nutrients stored in the liver and fatty tissues, with a diverse range of functions. Acyl-carnitines are involved in the transport and metabolism of fatty acids in and out of mitochondria, where they undergo beta oxidation.[55] Polyprenols and their phosphorylated derivatives also play important transport roles, in this case the transport of oligosaccharides across membranes. Polyprenol phosphate sugars and polyprenol diphosphate sugars function in extra-cytoplasmic glycosylation reactions, in extracellular polysaccharide biosynthesis (for instance, peptidoglycan polymerization in bacteria), and in eukaryotic protein N-glycosylation.[56][57] Cardiolipins are a subclass of glycerophospholipids containing four acyl chains and three glycerol groups that are particularly abundant in the inner mitochondrial membrane.[58] They are believed to activate enzymes involved with oxidative phosphorylation.[59]
[edit] Metabolism
The major dietary lipids for humans and other animals are animal and plant triglycerides, sterols, and membrane phospholipids. The process of lipid metabolism synthesizes and degrades the lipid stores and produces the structural and functional lipids characteristic of individual tissues.
[edit] Biosynthesis
In animals, when there is an oversupply of dietary carbohydrate, the excess carbohydrate is converted to triacylglycerol. This involves the synthesis of fatty acids from acetyl-CoA and the esterification of fatty acids in the production of triacylglycerol, a process called lipogenesis.[60] Fatty acids are made by fatty acid synthases that polymerize and then reduce acetyl-CoA units. The acyl chains in the fatty acids are extended by a cycle of reactions that add the acetyl group, reduce it to an alcohol, dehydrate it to an alkene group and then reduce it again to an alkane group. The enzymes of fatty acid biosynthesis are divided into two groups, in animals and fungi all these fatty acid synthase reactions are carried out by a single multifunctional protein,[61] while in plant plastids and bacteria separate enzymes perform each step in the pathway.[62][63] The fatty acids may be subsequently converted to triacylglycerols that are packaged in lipoproteins and secreted from the liver.
The synthesis of unsaturated fatty acids involves a desaturation reaction, whereby a double bond is introduced into the fatty acyl chain. For example, in humans, the desaturation of stearic acid by stearoyl-CoA desaturase-1 produces oleic acid. The doubly unsaturated fatty acid linoleic acid as well as the triply unsaturated î�-linolenic acid cannot be synthesized in mammalian tissues, and are therefore essential fatty acids and must be obtained from the diet.[64]
Triacylglycerol synthesis takes place in the endoplasmic reticulum by metabolic pathways in which acyl groups in fatty acyl-CoAs are transferred to the hydroxyl groups of glycerol-3-phosphate and diacylglycerol.[65]
Terpenes and isoprenoids, including the carotenoids, are made by the assembly and modification of isoprene units donated from the reactive precursors isopentenyl pyrophosphate and dimethylallyl pyrophosphate.[66] These precursors can be made in different ways. In animals and archaea, the mevalonate pathway produces these compounds from acetyl-CoA,[67] while in plants and bacteria the non-mevalonate pathway uses pyruvate and glyceraldehyde 3-phosphate as substrates.[66][68] One important reaction that uses these activated isoprene donors is steroid biosynthesis. Here, the isoprene units are joined together to make squalene and then folded up and formed into a set of rings to make lanosterol.[69] Lanosterol can then be converted into other steroids such as cholesterol and ergosterol.[69][70]
[edit] Degradation
Beta oxidation is the metabolic process by which fatty acids are broken down in the mitochondria and/or in peroxisomes to generate acetyl-CoA. For the most part, fatty acids are oxidized by a mechanism that is similar to, but not identical with, a reversal of the process of fatty acid synthesis. That is, two-carbon fragments are removed sequentially from the carboxyl end of the acid after steps of dehydrogenation, hydration, and oxidation to form a beta-keto acid, which is split by thiolysis. The acetyl-CoA is then ultimately converted into ATP, CO2, and H2O using the citric acid cycle and the electron transport chain.
Hence the Krebs Cycle can start at acetyl-CoA when fat is being broken down for energy if there is little or no glucose available.
The energy yield of the complete oxidation of the fatty acid palmitate is 106 ATP.[71] Unsaturated and odd-chain fatty acids require additional enzymatic steps for degradation.
[edit] Nutrition and health
Most of the lipid found in food is in the form of triacylglycerols, cholesterol and phospholipids. A minimum amount of dietary fat is necessary to facilitate absorption of fat-soluble vitamins (A, D, E and K) and carotenoids.[72] Humans and other mammals have a dietary requirement for certain essential fatty acids, such as linoleic acid (an omega-6 fatty acid) and alpha-linolenic acid (an omega-3 fatty acid) because they cannot be synthesized from simple precursors in the diet.[64] Both of these fatty acids are 18-carbon polyunsaturated fatty acids differing in the number and position of the double bonds. Most vegetable oils are rich in linoleic acid (safflower, sunflower, and corn oils). Alpha-linolenic acid is found in the green leaves of plants, and in selected seeds, nuts and legumes (particularly flax, rapeseed, walnut and soy).[73] Fish oils are particularly rich in the longer-chain omega-3 fatty acids eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA).[74] A large number of studies have shown positive health benefits associated with consumption of omega-3 fatty acids on infant development, cancer, cardiovascular diseases, and various mental illnesses, such as depression, attention-deficit hyperactivity disorder, and dementia.[75][76] In contrast, it is now well-established that consumption of trans fats, such as those present in partially hydrogenated vegetable oils, are a risk factor for cardiovascular disease.[77][78][79]
A few studies have suggested that total dietary fat intake is linked to an increased risk of obesity[80][81] and diabetes.[82][83] However, a number of very large studies, including the Women's Health Initiative Dietary Modification Trial, an eight year study of 49,000 women, the Nurses' Health Study and the Health Professionals Follow-up Study, revealed no such links.[84][85][86] None of these studies suggested any connection between percentage of calories from fat and risk of cancer, heart disease or weight gain. The Nutrition Source, a website maintained by the Department of Nutrition at the Harvard School of Public Health, summarizes the current evidence on the impact of dietary fat: "Detailed research'much of it done at Harvard'shows that the total amount of fat in the diet isn't really linked with weight or disease."[87]
[edit] See also
- Lipidomics
- Essential fatty acid
- Lipid microdomain
- Lipid signaling
- Protein-lipid interaction
- Lipoprotein
[edit] References
- ^ Stryer et al., p. 328.
- ^ Maitland, Jr Jones (1998). Organic Chemistry. W W Norton & Co Inc (Np). p. 139. ISBN 0-393-97378-6.
- ^ a b c Stryer et al., p. 330.
- ^ Fahy E, Subramaniam S, Brown HA, et al. (2005). "A comprehensive classification system for lipids". Journal of Lipid Research 46 (5): 839'61. doi:10.1194/jlr.E400004-JLR200. PMID 15722563.
- ^ Michelle A, Hopkins J, McLaughlin CW, Johnson S, Warner MQ, LaHart D, Wright JD (1993). Human Biology and Health. Englewood Cliffs, New Jersey, USA: Prentice Hall. ISBN 0-13-981176-1. OCLC 32308337.
- ^ Vance JE, Vance DE (2002). Biochemistry of Lipids, Lipoproteins and Membranes. Amsterdam: Elsevier. ISBN 0444511393. OCLC 51001207.
- ^ Brown HA, ed (2007). Lipodomics and Bioactive Lipids: Mass Spectrometry Based Lipid Analysis, Volume 432 (Methods in Enzymology). Boston: Academic Press. ISBN 0123738954. OCLC 166624879.
- ^ Hunt SM, Groff JL, Gropper SAS (1995). Advanced Nutrition and Human Metabolism. Belmont, CA: West Pub. Co. p. 98. ISBN 0-314-04467-1.
- ^ Devlin, pp. 193'95.
- ^ Hunter JE (November 2006). "Dietary trans fatty acids: review of recent human studies and food industry responses". Lipids 41 (11): 967'92. doi:10.1007/s11745-006-5049-y. PMID 17263298.
- ^ Fezza F, De Simone C, Amadio D, Maccarrone M (2008). "Fatty acid amide hydrolase: a gate-keeper of the endocannabinoid system". Subcellular Biochemistry 49: 101'32. doi:10.1007/978-1-4020-8831-5_4. PMID 18751909.
- ^ Coleman RA, Lee DP (2004). "Enzymes of triacylglycerol synthesis and their regulation". Progress in Lipid Research 43 (2): 134'76. doi:10.1016/S0163-7827(03)00051-1. PMID 14654091.
- ^ van Holde and Mathews, p. 630'31.
- ^ a b Hölzl G, Dörmann P (2007). "Structure and function of glycoglycerolipids in plants and bacteria". Progress in Lipid Research 46 (5): 225'43. doi:10.1016/j.plipres.2007.05.001. PMID 17599463. http://linkinghub.elsevier.com/retrieve/pii/S0163-7827(07)00015-X.
- ^ Honke K, Zhang Y, Cheng X, Kotani N, Taniguchi N (2004). "Biological roles of sulfoglycolipids and pathophysiology of their deficiency". Glycoconjugates Journal 21 (1'2): 59'62. doi:10.1023/B:GLYC.0000043749.06556.3d. PMID 15467400. http://www.kluweronline.com/art.pdf?issn=0282-0080&volume=21&page=59.
- ^ Farooqui AA, Horrocks LA, Farooqui T (2000). "Glycerophospholipids in brain: their metabolism, incorporation into membranes, functions, and involvement in neurological disorders". Chemistry and Physics of Lipids 106 (1): 1'29. doi:10.1016/S0009-3084(00)00128-6. PMID 10878232. http://linkinghub.elsevier.com/retrieve/pii/S0009-3084(00)00128-6. Retrieved 2009-04-12.
- ^ Ivanova PT, Milne SB, Byrne MO, Xiang Y, Brown HA (2007). "Glycerophospholipid identification and quantitation by electrospray ionization mass spectrometry". Methods in Enzymology 432: 21'57. doi:10.1016/S0076-6879(07)32002-8. PMID 17954212.
- ^ van Holde and Mathews, p. 844.
- ^ Paltauf F (1994). "Ether lipids in biomembranes". Chemistry and Physics of Lipids 74 (2): 101'39. doi:10.1016/0009-3084(94)90054-X. PMID 7859340.
- ^ Merrill AH, Sandhoff K. (2002). "Sphingolipids: metabolism and cell signaling",in New Comprehensive Biochemistry: Biochemistry of Lipids, Lipoproteins,and Membranes, Vance, D.E. and Vance, J.E., eds. Elsevier Science, NY. Ch. 14.
- ^ Devlin, pp. 421'22.
- ^ Hori T, Sugita M (1993). "Sphingolipids in lower animals". Prog. Lipid Res 32 (1): 25'45. doi:10.1016/0163-7827(93)90003-F. PMID 8415797.
- ^ Wiegandt H (1992). "Insect glycolipids". Biochimica et Biophysica Acta 1123 (2): 117'26. PMID 1739742.
- ^ Guan X, Wenk MR (2008). "Biochemistry of inositol lipids". Frontiers in Bioscience 13: 3239'51. doi:10.2741/2923. PMID 18508430.
- ^ Bach D, Wachtel E (2003). "Phospholipid/cholesterol model membranes: formation of cholesterol crystallites". Biochim Biophys Acta 1610 (2): 187'97. doi:10.1016/S0005-2736(03)00017-8. PMID 12648773.
- ^ Stryer et al., p. 749.
- ^ Bouillon R, Verstuyf A, Mathieu C, Van Cromphaut S, Masuyama R, Dehaes P, Carmeliet G (2006). "Vitamin D resistance". Best Practice & Research. Clinical Endocrinology & Metabolism 20 (4): 627'45. doi:10.1016/j.beem.2006.09.008. PMID 17161336. http://linkinghub.elsevier.com/retrieve/pii/S1521-690X(06)00079-0.
- ^ Russell DW (2003). "The enzymes, regulation, and genetics of bile acid synthesis". Annual Review of Biochemistry 72: 137'74. doi:10.1146/annurev.biochem.72.121801.161712. PMID 12543708.
- ^ Villinski JC, Hayes JM, Brassell SC, Riggert VL, Dunbar RB (2008). "Sedimentary sterols as biogeochemical indicators in the Southern Ocean". Organic Geochemistry 39 (5): 567'88. doi:10.1016/j.orggeochem.2008.01.009.
- ^ Deacon J (2005). Fungal Biology. Cambridge, MA: Blackwell Publishers. p. 342. ISBN 1-4051-3066-0.
- ^ Kuzuyama T, Seto H (2003). "Diversity of the biosynthesis of the isoprene units". Natural Product Reports 20 (2): 171'83. doi:10.1039/b109860h. PMID 12735695.
- ^ Rao AV, Rao LG (2007). "Carotenoids and human health". Pharmacological Research : the Official Journal of the Italian Pharmacological Society 55 (3): 207'16. doi:10.1016/j.phrs.2007.01.012. PMID 17349800. http://linkinghub.elsevier.com/retrieve/pii/S1043-6618(07)00035-7.
- ^ Brunmark A, Cadenas E (1989). "Redox and addition chemistry of quinoid compounds and its biological implications". Free Radical Biology & Medicine 7 (4): 435'77. doi:10.1016/0891-5849(89)90126-3. PMID 2691341.
- ^ Swiezewska E, Danikiewicz W (2005). "Polyisoprenoids: structure, biosynthesis and function". Progress in Lipid Research 44 (4): 235'58. doi:10.1016/j.plipres.2005.05.002. PMID 16019076. http://linkinghub.elsevier.com/retrieve/pii/S0163-7827(05)00022-6.
- ^ a b Raetz CR, Garrett TA, Reynolds CM, Shaw WA, Moore JD, Smith DC Jr, Ribeiro AA, Murphy RC,Ulevitch RJ, Fearns C, Reichart D, Glass CK, Benner C, Subramaniam S, Harkewicz R, Bowers-Gentry RC, Buczynski MW, Cooper JA, Deems RA, Dennis EA (2006). "Kdo2-Lipid A of Escherichia coli, a defined endotoxin that activates macrophages via TLR-4". Journal of Lipid Research 47 (5): 1097'111. doi:10.1194/jlr.M600027-JLR200. PMID 16479018.
- ^ Walsh CT (2004). "Polyketide and nonribosomal peptide antibiotics: modularity and versatility". Science 303 (5665): 1805'10. doi:10.1126/science.1094318. PMID 15031493.
- ^ Caffrey P, Aparicio JF, Malpartida F, Zotchev SB (2008). "Biosynthetic engineering of polyene macrolides towards generation of improved antifungal and antiparasitic agents". Current Topics in Medicinal Chemistry 8 (8): 639'53. doi:10.2174/156802608784221479. PMID 18473889. http://www.bentham-direct.org/pages/content.php?CTMC/2008/00000008/00000008/0003R.SGM. Retrieved 2009-04-12.
- ^ Minto RE, Blacklock BJ (2008). "Biosynthesis and function of polyacetylenes and allied natural products". Progress in Lipid Research 47 (4): 233'306. doi:10.1016/j.plipres.2008.02.002. PMID 18387369. PMC 2515280. http://linkinghub.elsevier.com/retrieve/pii/S0163-7827(08)00015-5. Retrieved 2009-04-12.
- ^ Stryer et al., pp. 329'331
- ^ Heinz E.(1996). Plant glycolipids: structure, isolation and analysis. in Advances in Lipid Methodology - 3, pp. 211'332 (ed. W.W. Christie, Oily Press, Dundee)
- ^ Stryer et al., pp. 333'34.
- ^ van Meer G, Voelker DR, Feigenson GW (2008). "Membrane lipids: where they are and how they behave". Nature Reviews. Molecular Cell Biology 9 (2): 112'24. doi:10.1038/nrm2330. PMID 18216768.
- ^ Feigenson GW (2006). "Phase behavior of lipid mixtures". Nature Chemical Biology 2 (11): 560'63. doi:10.1038/nchembio1106-560. PMID 17051225.
- ^ Wiggins PM (1990). "Role of water in some biological processes". Microbiological Reviews 54 (4): 432'49. PMID 2087221. PMC 372788. http://www.pubmedcentral.nih.gov/picrender.fcgi?artid=372788&blobtype=pdf.
- ^ Raschke TM, Levitt M (2005). "Nonpolar solutes enhance water structure within hydration shells while reducing interactions between them". Proceedings of the National Academy of Sciences U.S.A 102 (19): 6777'82. doi:10.1073/pnas.0500225102. PMID 15867152. PMC 1100774. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pubmed&pubmedid=15867152.
- ^ Brasaemle DL (December 2007). "Thematic review series: adipocyte biology. The perilipin family of structural lipid droplet proteins: stabilization of lipid droplets and control of lipolysis". J. Lipid Res 48 (12): 2547'59. doi:10.1194/jlr.R700014-JLR200. PMID 17878492. http://www.jlr.org/cgi/content/full/48/12/2547.
- ^ Stryer et al., p. 619.
- ^ Wang X (2004). "Lipid signaling". Current Opinions in Plant Biology 7 (3): 329'36. doi:10.1016/j.pbi.2004.03.012. PMID 15134755.
- ^ Eyster KM (2007). "The membrane and lipids as integral participants in signal transduction". Advances in Physiology Education 31 (1): 5'16. doi:10.1152/advan.00088.2006. PMID 17327576.
- ^ Hinkovska-Galcheva V, VanWay SM, Shanley TP, Kunkel RG (2008). "The role of sphingosine-1-phosphate and ceramide-1-phosphate in calcium homeostasis". Current Opinion in Investigational Drugs 9 (11): 1192'205. PMID 18951299.
- ^ Saddoughi SA, Song P, Ogretmen B (2008). "Roles of bioactive sphingolipids in cancer biology and therapeutics". Subcellular Biochemistry 49: 413'40. doi:10.1007/978-1-4020-8831-5_16. PMID 18751921.
- ^ Klein C, Malviya AN (2008). "Mechanism of nuclear calcium signaling by inositol 1,4,5-trisphosphate produced in the nucleus, nuclear located protein kinase C and cyclic AMP-dependent protein kinase". Frontiers in Bioscience 13: 1206'26. doi:10.2741/2756. PMID 17981624. http://www.bioscience.org/2008/v13/af/2756/fulltext.htm.
- ^ Boyce JA (2008). "Eicosanoids in asthma, allergic inflammation, and host defense". Current Molecular Medicine 8 (5): 335'49. doi:10.2174/156652408785160989. PMID 18691060. http://www.bentham-direct.org/pages/content.php?CMM/2008/00000008/00000005/0003M.SGM.
- ^ Be�towski J (2008). "Liver X receptors (LXR) as therapeutic targets in dyslipidemia". Cardiovascular Therapy 26 (4): 297'316. doi:10.1111/j.1755-5922.2008.00062.x. PMID 19035881.
- ^ Indiveri C, Tonazzi A, Palmieri F (October 1991). "Characterization of the unidirectional transport of carnitine catalyzed by the reconstituted carnitine carrier from rat liver mitochondria". Biochim. Biophys. Acta 1069 (1): 110'6. doi:10.1016/0005-2736(91)90110-T. PMID 1932043.
- ^ Parodi AJ, Leloir LF (April 1979). "The role of lipid intermediates in the glycosylation of proteins in the eucaryotic cell". Biochim. Biophys. Acta 559 (1): 1'37. doi:10.1016/0304-4157(79)90006-6. PMID 375981.
- ^ Helenius A, Aebi M (2001). "Intracellular functions of N-linked glycans". Science 291 (5512): 2364'69. doi:10.1126/science.291.5512.2364. PMID 11269317.
- ^ Gohil VM, Greenberg ML (2009). "Mitochondrial membrane biogenesis: phospholipids and proteins go hand in hand". Journal of Cell Biology 184 (4): 469'72. doi:10.1083/jcb.200901127. PMID 19237595. PMC 2654137. http://jcb.rupress.org/cgi/content/full/184/4/469.
- ^ Hoch FL (1992). "Cardiolipins and biomembrane function". Biochimica et Biophysica Acta 1113 (1): 71'133. PMID 10206472.
- ^ Stryer et al., p. 634.
- ^ Chirala S, Wakil S (2004). "Structure and function of animal fatty acid synthase". Lipids 39 (11): 1045'53. doi:10.1007/s11745-004-1329-9. PMID 15726818.
- ^ White S, Zheng J, Zhang Y (2005). "The structural biology of type II fatty acid biosynthesis". Annual Review of Biochemistry 74: 791'831. doi:10.1146/annurev.biochem.74.082803.133524. PMID 15952903.
- ^ Ohlrogge J, Jaworski J (1997). "Regulation of fatty acid synthesis". Annual Review of Plant Physiology and Plant Molecular Biology 48: 109'136. doi:10.1146/annurev.arplant.48.1.109. PMID 15012259.
- ^ a b Stryer et al., p. 643.
- ^ Stryer et al., pp. 733'39.
- ^ a b Kuzuyama T, Seto H (2003). "Diversity of the biosynthesis of the isoprene units". Natural Product Reports 20 (2): 171'83. doi:10.1039/b109860h. PMID 12735695.
- ^ Grochowski L, Xu H, White R (2006). "Methanocaldococcus jannaschii uses a modified mevalonate pathway for biosynthesis of isopentenyl diphosphate". Journal of Bacteriology 188 (9): 3192'98. doi:10.1128/JB.188.9.3192-3198.2006. PMID 16621811. PMC 1447442. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pubmed&pubmedid=16621811.
- ^ Lichtenthaler H (1999). "The 1-Dideoxy-D-xylulose-5-phosphate pathway of isoprenoid biosynthesis in plants". Annual Review of Plant Physiology and Plant Molecular Biology 50: 47'65. doi:10.1146/annurev.arplant.50.1.47. PMID 15012203.
- ^ a b Schroepfer G (1981). "Sterol biosynthesis". Annual Review of Biochemistry 50: 585'621. doi:10.1146/annurev.bi.50.070181.003101. PMID 7023367.
- ^ Lees N, Skaggs B, Kirsch D, Bard M (1995). "Cloning of the late genes in the ergosterol biosynthetic pathway of Saccharomyces cerevisiae'a review". Lipids 30 (3): 221'26. doi:10.1007/BF02537824. PMID 7791529.
- ^ Stryer et al., pp. 625'26.
- ^ Bhagavan, p. 903.
- ^ Russo GL (2009). "Dietary n-6 and n-3 polyunsaturated fatty acids: from biochemistry to clinical implications in cardiovascular prevention". Biochemical Pharmacology 77 (6): 937'46. doi:10.1016/j.bcp.2008.10.020. PMID 19022225. http://linkinghub.elsevier.com/retrieve/pii/S0006-2952(08)00777-6.
- ^ Bhagavan, p. 388.
- ^ Riediger ND, Othman RA, Suh M, Moghadasian MH (2009). "A systemic review of the roles of n-3 fatty acids in health and disease". Journal of the American Dietetic Association 109 (4): 668'79. doi:10.1016/j.jada.2008.12.022. PMID 19328262.
- ^ Galli C, Risé P (2009). "Fish consumption, omega 3 fatty acids and cardiovascular disease. The science and the clinical trials". Nutrition and Health (Berkhamsted, Hertfordshire) 20 (1): 11'20. PMID 19326716.
- ^ Micha R, Mozaffarian D (2008). "Trans fatty acids: effects on cardiometabolic health and implications for policy". Prostaglandins, Leukotrienes, and Essential Fatty Acids 79 (3'5): 147'52. doi:10.1016/j.plefa.2008.09.008. PMID 18996687.
- ^ Dalainas I, Ioannou HP (2008). "The role of trans fatty acids in atherosclerosis, cardiovascular disease and infant development". International Angiology: a Journal of the International Union of Angiology 27 (2): 146'56. PMID 18427401.
- ^ Mozaffarian D, Willett WC (2007). "Trans fatty acids and cardiovascular risk: a unique cardiometabolic imprint?". Current Atherosclerosis Reports 9 (6): 486'93. doi:10.1007/s11883-007-0065-9. PMID 18377789.
- ^ Astrup A, Dyerberg J, Selleck M, Stender S (2008). "Nutrition transition and its relationship to the development of obesity and related chronic diseases". Obesity Review 9 Suppl 1: 48'52. doi:10.1111/j.1467-789X.2007.00438.x. PMID 18307699.
- ^ Astrup A (2005). "The role of dietary fat in obesity". Seminars in Vascular Medicine 5 (1): 40'47. doi:10.1055/s-2005-871740. PMID 15968579.
- ^ Ma Y. et al (2006). "Low-carbohydrate and high-fat intake among adult patients with poorly controlled type 2 diabetes mellitus". Nutrition 22 (11-12): 1129'1136. doi:10.1016/j.nut.2006.08.006. PMID 17027229.
- ^ Astrup A (2008). "Dietary management of obesity". JPEN Journal of Parenteral and Enteral Nutrition 32 (5): 575'77. doi:10.1177/0148607108321707. PMID 18753397. http://pen.sagepub.com/cgi/pmidlookup?view=long&pmid=18753397. Retrieved 2009-04-12.
- ^ Beresford SA, Johnson KC, Ritenbaugh C, et al. (2006). "Low-fat dietary pattern and risk of colorectal cancer: the Women's Health Initiative Randomized Controlled Dietary Modification Trial". JAMA: the Journal of the American Medical Association 295 (6): 643'54. doi:10.1001/jama.295.6.643. PMID 16467233.
- ^ Howard BV, Manson JE, Stefanick ML, et al. (2006). "Low-fat dietary pattern and weight change over 7 years: the Women's Health Initiative Dietary Modification Trial". JAMA: the Journal of the American Medical Association 295 (1): 39'49. doi:10.1001/jama.295.1.39. PMID 16391215. http://jama.ama-assn.org/cgi/pmidlookup?view=long&pmid=16391215.
- ^ Howard BV, Van Horn L, Hsia J, et al. (2006). "Low-fat dietary pattern and risk of cardiovascular disease: the Women's Health Initiative Randomized Controlled Dietary Modification Trial". JAMA : the Journal of the American Medical Association 295 (6): 655'66. doi:10.1001/jama.295.6.655. PMID 16467234.
- ^ "Fats and Cholesterol: Out with the Bad, In with the Good - What Should You Eat? - The Nutrition Source - Harvard School of Public Health". http://www.hsph.harvard.edu/nutritionsource/what-should-you-eat/fats-full-story/index.html. Retrieved 2009-05-12.
[edit] Bibliography
- Bhagavan NV (2002). Medical Biochemistry. San Diego: Harcourt/Academic Press. ISBN 0-12-095440-0. http://books.google.com/?id=vT9YttFTPi0C&printsec=frontcover.
- Devlin TM (1997). Textbook of Biochemistry: With Clinical Correlations (4th ed.). Chichester: John Wiley & Sons. ISBN 0-471-17053-4.
- Stryer L, Berg JM, Tymoczko JL (2007). Biochemistry (6th ed.). San Francisco: W.H. Freeman. ISBN 0-7167-8724-5.
- Van Holde KE, Mathews CK (1996). Biochemistry (2nd ed.). Menlo Park, Calif: Benjamin/Cummings Pub. Co. ISBN 0-8053-3931-0.
[edit] External links
| Wikimedia Commons has media related to: Lipids |
Introductory
- List of lipid-related web sites
- Nature Lipidomics Gateway - Round-up and summaries of recent lipid research
- Lipid Library - General reference on lipid chemistry and biochemistry
- Cyberlipid.org - Resources and history for lipids.
- Molecular Computer Simulations - Modeling of Lipid Membranes
- Lipids, Membranes and Vesicle Trafficking - The Virtual Library of Biochemistry and Cell Biology
Nomenclature
Databases
- LIPID MAPS - Comprehensive lipid and lipid-associated gene/protein databases.
- LipidBank - Japanese database of lipids and related properties, spectral data and references.
- LIPIDAT - Database composed mainly of phospholipids and associated thermodynamic data.
General
- ApolloLipids - Provides dyslipidemia and cardiovascular disease prevention and treatment information as well as continuing medical education programs
- National Lipid Association - Professional medical education organization for health care professionals who seek to prevent morbidity and mortality stemming from dyslipidemias and other cholesterol-related disorders.
|
|||||||||||||||||||||||||||||||||||||
|
|||||
|
||||||||||||||||||||
|
||||||||||||||||||||||
|
|||||||||||
|
|||||||||||||||||||||||||||
|
SOURCES.COM is an online portal and directory for journalists, news media, researchers and anyone seeking experts, spokespersons, and reliable information resources. Use SOURCES.COM to find experts, media contacts, news releases, background information, scientists, officials, speakers, newsmakers, spokespeople, talk show guests, story ideas, research studies, databases, universities, associations and NGOs, businesses, government spokespeople. Indexing and search applications by Ulli Diemer and Chris DeFreitas.
For information about being included in SOURCES as a expert or spokesperson see the FAQ . For partnerships, content and applications, and domain name opportunities contact us.